| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-07-13 19:49:05 UTC |
|---|
| Update Date | 2016-07-20 21:00:34 UTC |
|---|
| Lmdb | LMDB00404 |
|---|
| Secondary Accession Numbers | None |
|---|
| Metabolite Identification |
|---|
| Common Name | Galacturonic acid |
|---|
| Description | Galacturonic acid, also known as D-galacturonate or sodium pectate, belongs to the class of organic compounds known as glucuronic acid derivatives. Glucuronic acid derivatives are compounds containing a glucuronic acid moiety (or a derivative), which consists of a glucose moiety with the C6 carbon oxidized to a carboxylic acid. Galacturonic acid exists in all living species, ranging from bacteria to plants to humans. Galacturonic acid is found, on average, in the highest concentration within flaxseeds (Linum usitatissimum) and grape wine. Galacturonic acid has also been detected, but not quantified in, several different foods, such as olives (Olea europaea), milk (cow), domestic pigs (Sus scrofa domestica), german camomiles (Matricaria recutita), and figs (Ficus carica). This could make galacturonic acid a potential biomarker for the consumption of these foods. Galacturonic acid is a primary metabolite. Primary metabolites are metabolically or physiologically essential metabolites. They are directly involved in an organism’s growth, development or reproduction. Based on a literature review a significant number of articles have been published on Galacturonic acid. |
|---|
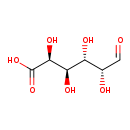
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2S,3R,4S,5R)-2,3,4,5-Tetrahydroxy-6-oxohexanoic acid | ChEBI | | D-Galacturonic acid | ChEBI | | (2S,3R,4S,5R)-2,3,4,5-Tetrahydroxy-6-oxohexanoate | Generator | | D-Galacturonate | Generator | | Galacturonate | Generator | | DL-Galacturonic acid | HMDB | | D-Galactopyranuronic acid | HMDB | | Galacturonic acid, (D)-isomer | HMDB | | Galacturonic acid, (alpha-D)-isomer | HMDB | | Galacturonic acid, calcium, sodium salt, (D)-isomer | HMDB | | Galacturonic acid, monosodium salt, (D)-isomer | HMDB | | Aldehydo-D-galacturonate | HMDB | | Polygalacturonic acid, aluminum salt | HMDB | | Sodium pectate | HMDB | | Galacturonan | HMDB | | Homogalacturonan | HMDB | | Pectic acid | HMDB | | Polygalacturonic acid homopolymer | HMDB | | Polygalacturonic acid, sulfated | HMDB | | Calcium polygalacturonate | HMDB | | Pectate | HMDB | | Polygalacturonic acid | HMDB | | Polygalacturonic acid, calcium salt | HMDB | | Polygalacturonic acid, homopolymer sodium salt | HMDB | | Sodium polygalacturonate | HMDB | | Anhydrogalacturonic acid | HMDB | | Calcium pectate | HMDB | | Polygalacturonic acid, homopolymer (D)-isomer | HMDB | | Aldehydo-D-galacturonic acid | HMDB | | (DL)-Galacturonic acid | HMDB | | (D)-Galacturonic acid | HMDB | | Galacturonic acid | MeSH |
|
|---|
| Chemical Formula | C6H10O7 |
|---|
| Average Molecular Weight | 194.1394 |
|---|
| Monoisotopic Molecular Weight | 194.042652674 |
|---|
| IUPAC Name | (2S,3R,4S,5R)-2,3,4,5-tetrahydroxy-6-oxohexanoic acid |
|---|
| Traditional Name | aldehydo-D-galacturonic acid |
|---|
| CAS Registry Number | 14982-50-4 |
|---|
| SMILES | O[C@@H](C=O)[C@@H](O)[C@@H](O)[C@H](O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C6H10O7/c7-1-2(8)3(9)4(10)5(11)6(12)13/h1-5,8-11H,(H,12,13)/t2-,3+,4+,5-/m0/s1 |
|---|
| InChI Key | IAJILQKETJEXLJ-RSJOWCBRSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as glucuronic acid derivatives. Glucuronic acid derivatives are compounds containing a glucuronic acid moiety (or a derivative), which consists of a glucose moiety with the C6 carbon oxidized to a carboxylic acid. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Glucuronic acid derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Glucuronic acid or derivatives
- Hexose monosaccharide
- Medium-chain hydroxy acid
- Medium-chain fatty acid
- Beta-hydroxy acid
- Hydroxy fatty acid
- Monosaccharide
- Fatty acyl
- Hydroxy acid
- Fatty acid
- Alpha-hydroxy acid
- Beta-hydroxy aldehyde
- Alpha-hydroxyaldehyde
- Secondary alcohol
- Polyol
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Aldehyde
- Carbonyl group
- Alcohol
- Organic oxide
- Hydrocarbon derivative
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Ontology |
|---|
| Status | Detected but not Quantified |
|---|
| Origin | Not Available |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-000i-9800000000-1d25ab641e968717daf1 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (5 TMS) - 70eV, Positive | splash10-00n0-3139450000-1762a4c61557c689a607 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 10V, Positive (Annotated) | splash10-03e9-0900000000-04b6c8cb4b4b2c077202 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 25V, Positive (Annotated) | splash10-0lk9-9700000000-4260c2e124c927041b62 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - Quattro_QQQ 40V, Positive (Annotated) | splash10-004i-9000000000-650b5bb9db964b1c3e87 | Spectrum | | LC-MS/MS | LC-MS/MS Spectrum - , negative | splash10-000i-9410000000-180d0e747dbcb5b21b01 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-056s-2900000000-86606b8382ffe403bc8e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-9500000000-af67bc97552641417bee | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-9100000000-205a7cff1e298283a080 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-05p9-9700000000-ee070fab487c9f3e49b7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4r-9400000000-284825e422ba9585f17b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9100000000-64d4f9d758e2a4eeba68 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01y5-7900000000-df218a4e340a0ff668c1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03kl-9100000000-d22d4d2644d77203331d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-08fu-9000000000-93301767b758d212114f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00fr-9300000000-77aa1619935fe785e397 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a6r-9000000000-da4be7e32a829503c021 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a6r-9000000000-c1c9308cd925f43d015f | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 2D NMR | [1H,13C] 2D NMR Spectrum | Not Available | Spectrum |
|
|---|