| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-08-01 08:05:18 UTC |
|---|
| Update Date | 2016-08-01 19:18:54 UTC |
|---|
| Lmdb | LMDB00923 |
|---|
| Secondary Accession Numbers | None |
|---|
| Metabolite Identification |
|---|
| Common Name | Vinylbital |
|---|
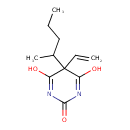
| Description | Vinylbital, also known as vinylbitone, belongs to the class of organic compounds known as barbituric acid derivatives. Barbituric acid derivatives are compounds containing a perhydropyrimidine ring substituted at C-2, -4 and -6 by oxo groups. Vinylbital is an extremely weak basic (essentially neutral) compound (based on its pKa). It was developed by Aktieboleget Pharmacia in the 1950s. |
|---|
| Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Vinylbitone | Kegg | | 5- Vinyl-5-(1-methylbutyl)barbituric acid | MeSH | | Bykonox | MeSH | | Speda | MeSH | | Suppoptanox | MeSH | | Butylvinal | MeSH | | Butylvinal, (-)-isomer | MeSH |
|
|---|
| Chemical Formula | C11H16N2O3 |
|---|
| Average Molecular Weight | 224.26 |
|---|
| Monoisotopic Molecular Weight | 224.116092383 |
|---|
| IUPAC Name | 5-ethenyl-4,6-dihydroxy-5-(pentan-2-yl)-2,5-dihydropyrimidin-2-one |
|---|
| Traditional Name | speda |
|---|
| CAS Registry Number | Not Available |
|---|
| SMILES | CCCC(C)C1(C=C)C(O)=NC(=O)N=C1O |
|---|
| InChI Identifier | InChI=1S/C11H16N2O3/c1-4-6-7(3)11(5-2)8(14)12-10(16)13-9(11)15/h5,7H,2,4,6H2,1,3H3,(H2,12,13,14,15,16) |
|---|
| InChI Key | KGKJZEKQJQQOTD-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as barbituric acid derivatives. Barbituric acid derivatives are compounds containing a perhydropyrimidine ring substituted at C-2, -4 and -6 by oxo groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Diazines |
|---|
| Sub Class | Pyrimidines and pyrimidine derivatives |
|---|
| Direct Parent | Barbituric acid derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Barbiturate
- N-acyl urea
- Ureide
- 1,3-diazinane
- Dicarboximide
- Urea
- Carbonic acid derivative
- Carboxylic acid derivative
- Azacycle
- Organic nitrogen compound
- Organonitrogen compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Carbonyl group
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Ontology |
|---|
| Status | Detected but not Quantified |
|---|
| Origin | Not Available |
|---|
| Biofunction | Not Available |
|---|
| Application | Not Available |
|---|
| Cellular locations | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Experimental Properties | | Property | Value | Reference |
|---|
| Melting Point | Not Available | Not Available | | Boiling Point | Not Available | Not Available | | Water Solubility | Not Available | Not Available | | LogP | Not Available | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4l-6910000000-45c002b8e7e2cacf8e9c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-1290000000-480bd7293d5cb6e08fe7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0zfr-1910000000-8480c995790aa1d3e440 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014l-9100000000-c387c0d7efcf6599e21f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-007o-8960000000-1af46c2654fd3c1501ee | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-9710000000-05382ed47f03ae9fca94 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000x-9500000000-dade22662680d6b7f7ba | Spectrum |
|
|---|